APPLICATION DOMAINS

APPLICATION DOMAINS
Integration of photonics devices tailored for different pilot-cases is a complex challenge comprising several photonics sub-technologies. To address this challenge, PhotonMed builds on cross-disciplinary value chains to advance the uptake of photonics technologies in in vitro diagnostics, in vivo diagnostics and personalized monitoring domains.
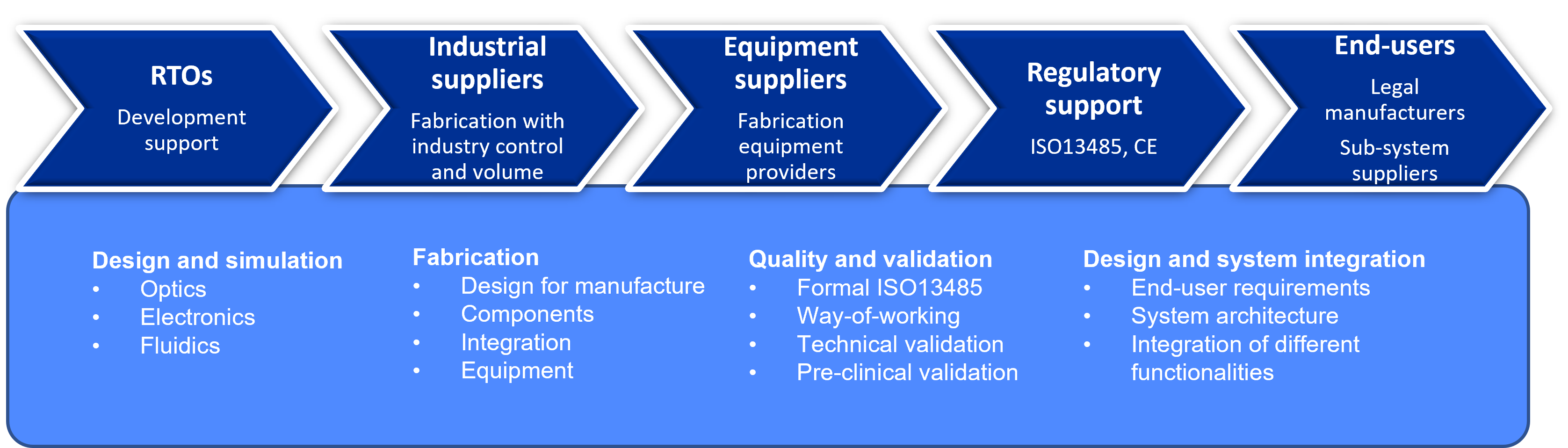
Value chains engage the key players, with complementing capabilities to accelerate and streamline the market entry of photonics medical devices.
- RTOs provide development support
- industrial suppliers advance their capability for high-volume fabrication with industrial control.
- Equipment suppliers provide fabrication equipment for the production of photonics devices.
- Regulatory support covers ISO13485 dealing with the manufacture and CE covering holistically regulations in medical devices.
- End-users provide commercialization path and manufacturing ecosystem for the pilot-cases.
All the technologies are validated in application relevant pilot cases.
PhotonMed pilot cases are classified into the following thematic application domains.
In vitro
In vitro diagnostics (IVD) refers to tests performed on samples such as blood or tissue that have been taken from the human body. These tests are conducted outside the body, typically in a laboratory setting, to detect diseases, conditions, or infections. Point-of-care testing (POCT), on the other hand, refers to medical diagnostic testing performed at or near the point of care, that is, at the time and place of patient care.
Impact
- Better diagnostic efficiency
- Reduce healthcare costs
- Provide clear benefits for well-being and quality of primary healthcare for patients

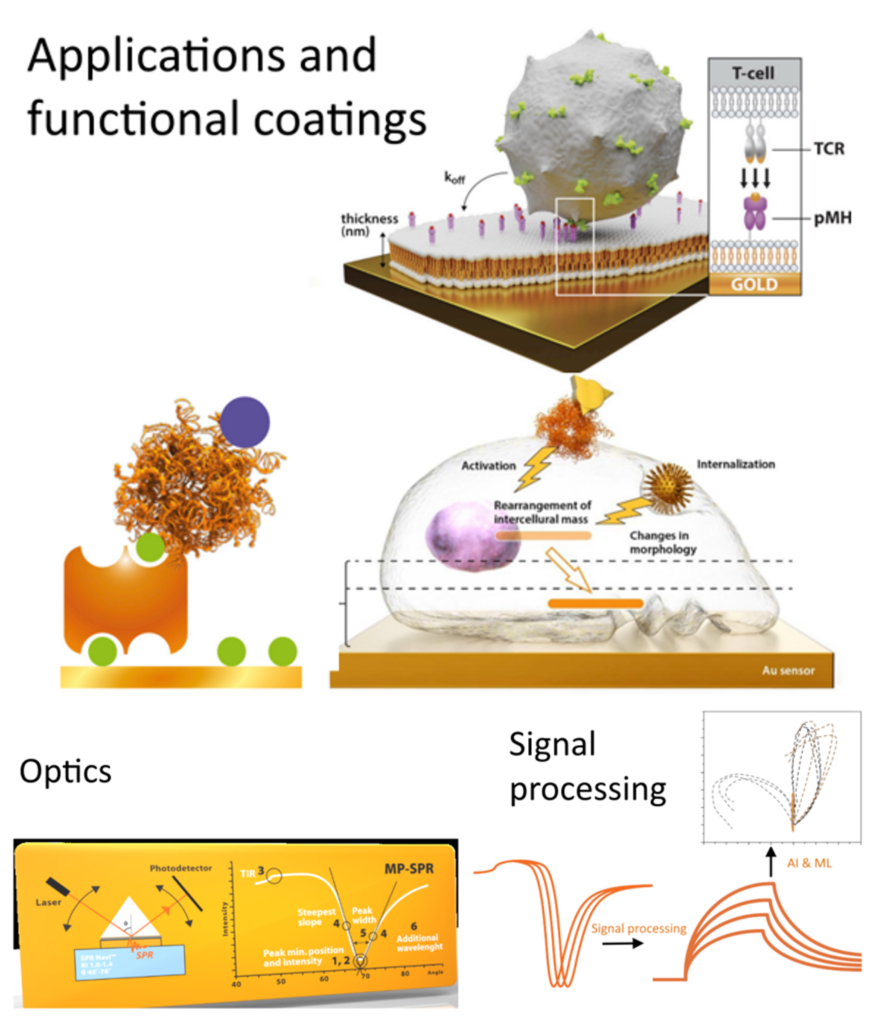
Multiparametric SPR for measurement of native cells
Bionavis will enhance MP-SPR capabilities by introducing advanced signal processing and AI-assisted analysis to identify key curve regions, automate evaluation and improve data quality, while updating the software with a user-friendly interface tailored for biological and clinical users. In addition, consortium expertise in fluidics, optics, and mechanics will reduce pump- and valve-induced fluctuations and optimize laser configurations, increasing sensitivity and reliability.
Objectives:
- Transform MP-SPR into a more powerful, accurate, and user-oriented tool, accelerating drug development, improving research quality, and expanding its adoption in new biological and clinical applications.
Partners:
- University of Freiburg, VTT

Multimodal blood diagnostics for point-of-care application
Bialoom is on a mission to accelerate diagnosis and therapy of acute blood infections and sepsis. Sepsis is a deadly health complication of infections that can result from antimicrobial resistance of pathogens. Fast diagnosis will be achieved by enabling multi-functional lab-quality blood tests next to the critically ill patient (point of care) at a small fraction of the time and cost of traditional molecular tests and blood cultures.
Objectives:
- Streamline sensor design and fabrication → >90% yield in final foundry-compatible devices
- Prepare for hyperplexed tests (towards 50 tests/cartridge)
- Simultaneous protein and pathogen detection from total blood samples in <30 mins
Partners:
- AMO, CyRIC, University of Freiburg, Scienion, TNO, VTT


DNA extraction from urine for PoC diagnostics
Qurin is developing a fast, robust, and finally PoC-usable urine sample preparation method to concentrate cfDNA and gDNA up to 1000x. Research will optimize urine collection (void/fraction) and extraction to enable reducing sample volume from ~400 mL to ~100 µL, compatible with optical sensing platforms. While primarily aimed at bladder cancer detection, the method could also support broader applications such as STD, UTI, and antibiotic resistance testing.
Objectives:
- Develop of a RUO kit to demonstrate the technology in first studies within this consortium, working together with various diagnostic platform developers in PhotonMed.
Partners:
Surfix, Bialoom, MEEP


Photonic IVD platform
It is Surfix’s mission to empower healthcare providers and patients worldwide with an advanced point-of-care (POC) platform that makes low-abundance biomarker testing accessible, delivering earlier insights, more accurate diagnoses, and better disease management. To this end, Surfix is developing an in-vitro diagnostics (IVD) platform based on photonic biochips. The platform consists of a desktop reader and microfluidic cartridges and enables simultaneous detection of multiple biomarkers with lab-level performance in a POC environment.
Objectives
- In PhotonMed, Surfix is advancing and upscaling several technologies and processes to make the step from a RUO system to an IVDR certified diagnostic platform.
Partners
- CSEM, Scienion

Photonic IVD platform for home use
MEEP develops an at-home minilab that allows individuals to have on-the-spot access to key health parameters. The product exists out of a reusable reader, a disposable test and a smartphone application. Powered by patented silicon photonics technology developed over more than a decade, MEEP is redefining how high-quality diagnostics can be delivered beyond the laboratory.
Objectives:
- Meep aims to increase the clinical and commercial value of the CRP test by demonstrating a high-sensitivity CRP assay intended for monitoring patients with cardiovascular disease.
- Elevate the Meep in vitro diagnostic platform via sensitivity optimization, pilot-line production readiness, and the development of a plasma sampling system compatible with decentralized at-home testing.
- Partners: IMEC, Voxdale, Scienion


NIR/MIR spectroscopy for glucose monitoring in organoids
Femtoprint will develop an optofluidic device for an innovative glucose monitoring of Human organoid cultures in microfluidic organ-on-chip (OoC) systems applications and demonstrate its functionality on a Liver-Pancreas-Assay by monitoring glucose concentration in cell culture compartment of the OoC.
Objectives:
- Identify and integrate near-infrared (NIR)/mid-infrared (MIR) transparent materials which are suitable for in vitro applications in OoC devices.
- Design and assemble a NIR/MIR demonstrator for glucose monitoring in OoC devices.
Impact:
- First non-invasive, real-time methods glucose monitoring in multi-organ chips
Partners:
- Alpes Lasers, CSEM
In vivo
In vivo diagnostics refers to diagnostic procedures performed within a living organism. In vivo diagnostics covers a broad field of methods including, for example, endoscopy and medical imaging. These procedures are crucial for providing real-time insights into the body’s internal structures and functions, allowing for accurate diagnosis and monitoring of various conditions.
Impact
- Improve healthcare outcomes
- Improve the quality of live for patients
- Reduce reoperations
- Reduce healthcare costs
- Accelerate economic growth


Integrated Swept Source Optical Coherence Tomography
Scinvivo’s Lumante Optical Coherence Tomography (OCT) imaging system helps urologists characterize bladder cancer during surgical procedures. It provides real-time, high-resolution cross-sectional imaging via a MEMS-scanned catheter. This enables clinicians to visualize microstructural changes in the bladder wall intraoperatively, supporting earlier detection, more accurate staging, and more personalized therapy—within a standard endoscopic workflow.
Objective:
- Develop a miniaturised, fibre-coupled SS-OCT module based on integrated photonics with on-chip source, interferometer and detection.
Partners:
- Ligentec, III-V Lab, CSEM, Philips

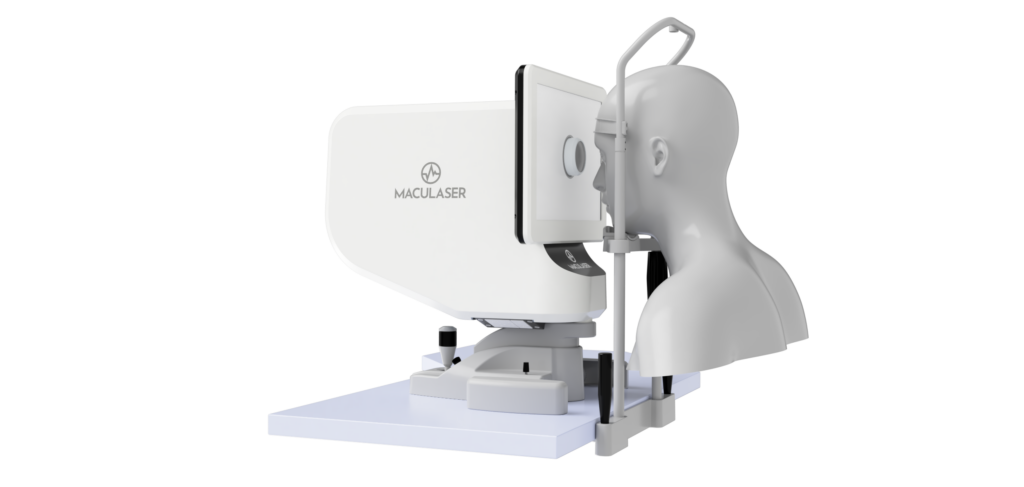
Ophthalmic laser treatment system with retinal temperature control
Maculaser is developing an advanced ophthalmic laser system for age-related macular degeneration and diabetic retinopathy that delivers real-time, patient-specific thermal dosing using retinal temperature feedback. An 810 nm laser, recording apparatus for temperature determination, and fundus/pupil imaging are integrated in an optomechanical platform to enable closed-loop laser-power control to activate cellular heat-shock response without retinal damage.
Objectives:
- Developing and testing new features for the laser treatment platform
- improved pupil imaging, improved fundus imaging, integration to the platform
- Developing a patient interface for retinal temperature determination
- Modeling light propagation in the eye
- Sophisticated eye model for R&D purposes
Partners:
- VTT, TNO

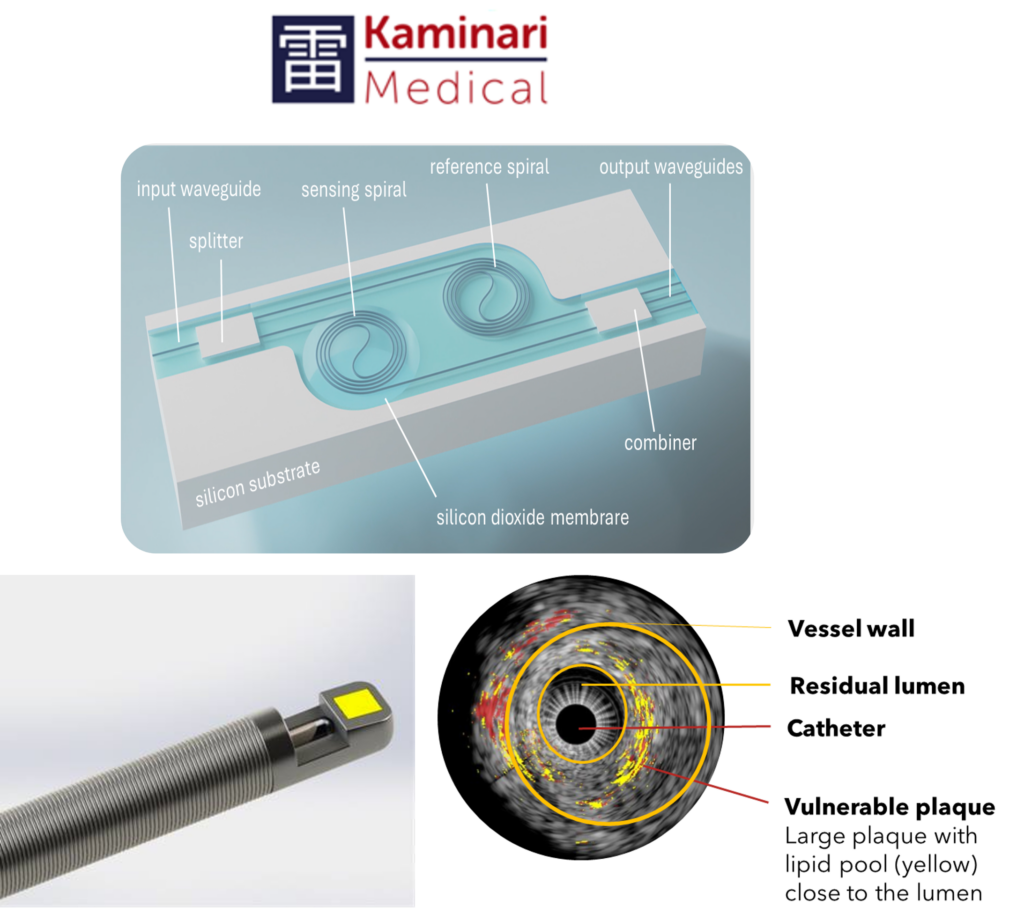
Intravascular lipid imaging with light ultrasound
The Kaminari intravascular ultrasound and photo acoustic (IVPA) system is a dual modality intracoronary imaging device. Intravascular ultrasound (IVUS) and photoacoustic (PA) modalities are combined in one device for simultaneous acquisition and co-registration of vessel structure and plaque composition data. The KAM IVPA system consists of a disposable intravascular ultrasound (IVUS) catheter with a photoacoustic (PA) component, a console and a pullback device. The system is to be used in the cardiac catheterization laboratory (cathlab).
Objectives:
- Realize an IPUT based (Integrated Photonic Ultrasound Transducer) ultrasound detector with an extreme bandwidth of 1-10 MHz, a NEP (Noise Equivalent Pressure) below 3 Pa and a sensing aperture width < 0.5 mm that could fit in a IVPA catheter.
- KAM will demonstrate the added value of combining IVUS and IVPA as an improved clinical decision support tool by developing a FUMO to be used in a FIH study at Erasmus MC.
Innovation:
- Intravascular lipid imaging with light and ultrasound
Partners:
- VTT, TNO, Erasmus University, IMEC, Philips
Micro imager for a catheter for in-vivo surgical guidance
Ficontec will develop an automated packaging process to enable reliable and scalable production of miniaturized optical probes. Using input from Tyndall National Institute’s prototypes and test results, ficonTEC will develop the manufacturing process, along with integrating the probes part-by-part to ensure the project’s output meet all quality requirements. These manufacturing process of the probes can then be adopted by an ISO13485 certified facility to support its production for preclinical trials.
Objectives:
- Enabling different types of diagnostic measurements through multimodal sensing using the imaging sensor and application-specific illumination.
- Allowing for deeper imaging within the human body, as we aim to further miniaturize the footprint of catheters with TNI’s imager design.
- Enabling a camera-on-catheter technology platform which opens up the potential for application specific designs.
- Enabling hardware specific image analytics being developed by TNI for e.g. feature segmentation and image enhancement.
Partners:
- FemtoPrint, Tyndall National Institute

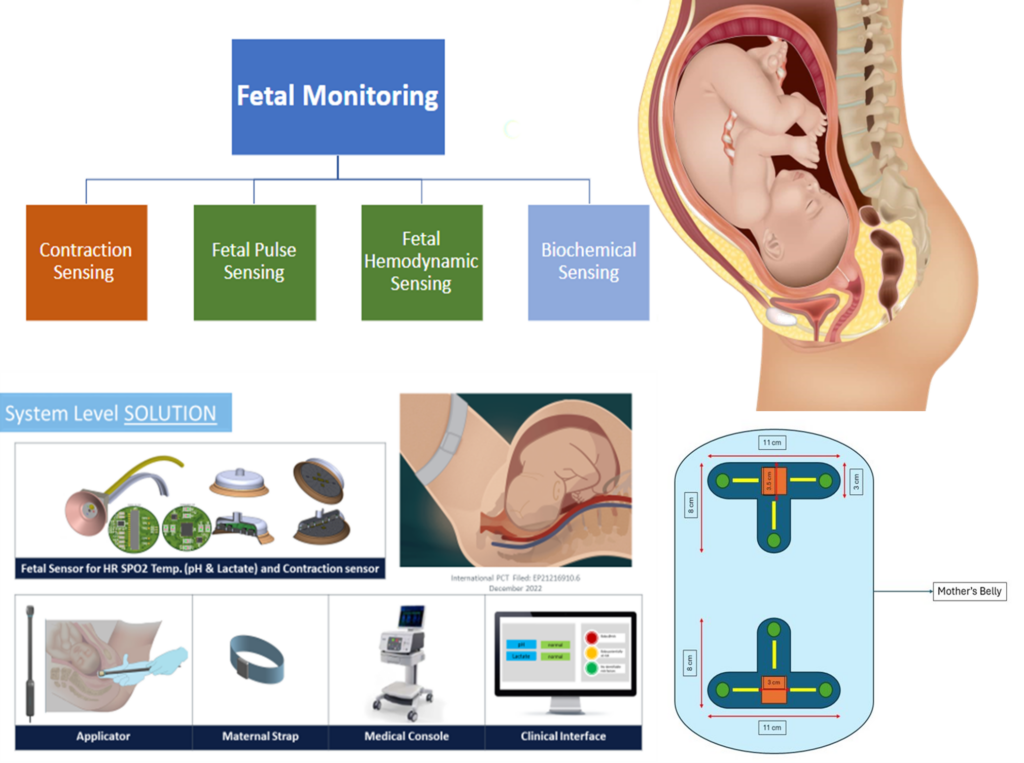
Hypoxia Monitoring for Acute Medical Applications and Intrapartum Care
Sanmina designs, manufactures, and repairs some of the world’s most complex and innovative optical, electronic, and mechanical products. In the PhotonMed project, Sanmina plays a key role in supporting the in-vivo device research pilot, and the manufacturing and validation of pilot case for hypoxia sensing device in acute medical applications, including intrapartum monitoring.
As a world-class contract electronics manufacturer (CEM), Sanmina is responsible for device manufacturing and assembly within a GMP and ISO 13485-certified environment. Working closely with Tyndall National Institute, Sanmina contributes critical expertise in Quality Management Systems (QMS) and Design for Manufacturing (DFM) to ensure the reliability, scalability, and clinical readiness of the technology.
Objective:
- Developing and testing a prototype non-invasive medical sensor platform designed to monitor vital signs and help prevent hypoxia in clinical settings
Partners:
- Sanmina, Tyndall National Institute
Personalized Monitoring
Personalized wearable monitoring refers to the use of wearable devices that can continuously monitor various physiological parameters and provide personalized health insights. These devices can measure a range of data, including heart rate, oxygen levels, and other vital signs, to offer a comprehensive view of an individual’s health.
Impact
- Promotes a healthy lifestyle
- Enables homecare opportunities
- Reduces healthcare costs
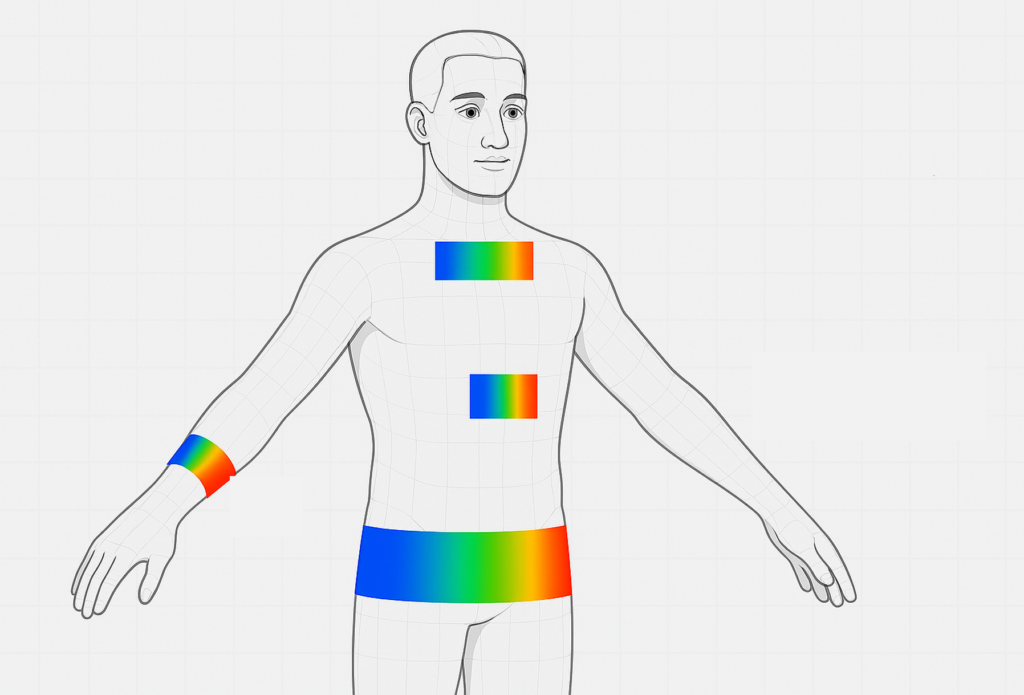
VIS/MIR spectral detection in wearables
In Polar’s use case, partners develop lasers and photonic modules for personalized monitoring at wavelengths covering from VIS (visible) to MIR (mid-infrared) range. Conventionally optical wearable devices utilize either single or two wavelength LEDs to track heart rate and SpO2 signals at visible and near-infrared (NIR) range. In stationary conditions, these are highly reliable and a focus in research of narrow band systems has been to increase reliability either using different form factors or improving the signal processing.
Objectives:
- Research on discrete laser sources that are integrated with light combiner elements.
- Research on miniaturized integration of photonic modules for personalised monitoring at wavelengths covering from VIS to MIR range.
- Wearable platform for laser testing and characterization and analytical tool development for data analysis.
- Tissue Optics Monte Carlo Analysis: investigation of the influence of skin types, gender, age, ethnicity etc.
- Demonstrator concepting. Dehydration monitoring is used as an example application.
Partners:
- POL, IMEC, VTT, III-V Lab, Wisematic, University of Tampere, Alpes Lasers, TNO

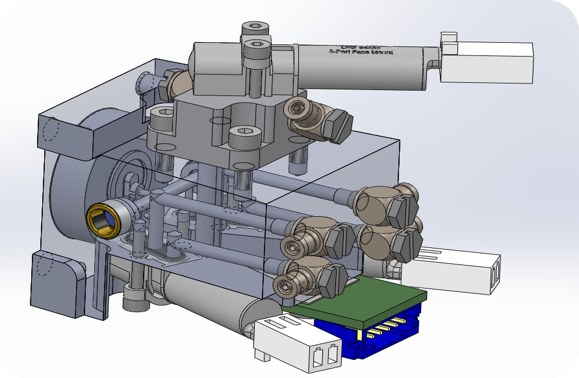
Photonic gas sensor for breath analysis
Gasera’s patented cantilever-enhanced photoacoustic technology is among the most sensitive optical gas sensing methods, achieving record sensitivity with an 8 cm path length in a simple cell. However, the 8 cm cell (4 mm diameter) is still too large for embedded or wearable sensors, and the current sample-and-hold mode limits continuous measurements, such as exhaled breath.
Objective:
- To miniaturize ultra-sensitive cantilever-enhanced photoacoustic technology into a continuous-flow platform with a sensor cell under 10 mm long and 1–2 mm in diameter. The platform’s versatility allows detection of diverse molecules using light sources from visible to far-infrared.
Innovation:
- Ultra-sensitive gas sensor platform for continuous breath analysis.
Partners:
- University of Tampere, VTT, MicroTEC, University of Freiburg


Light therapy device
CUROLED develops an innovative pilot application for photomedical smart textile wearables. The project focuses on enabling technologies for flexible LED-based therapy systems designed to support joint and muscular health. It establishes and validates a scalable platform for seamless embedded LED integration into textile substrates, advancing the reliability of flexible and stretchable photonic systems and forming the technological foundation for next-generation photomedical wearables in the healthcare market.
Objectives:
- Development, manufacturing and validation of personalized phototherapeutic wearable prototypes
- Development and advancement of technologies for integrating LEDs and sensors into flexible textile substrates.
- Verification of prototype effectiveness through targeted application tests
Partners:
VTT, TNO
